Agrandissement : Illustration 1
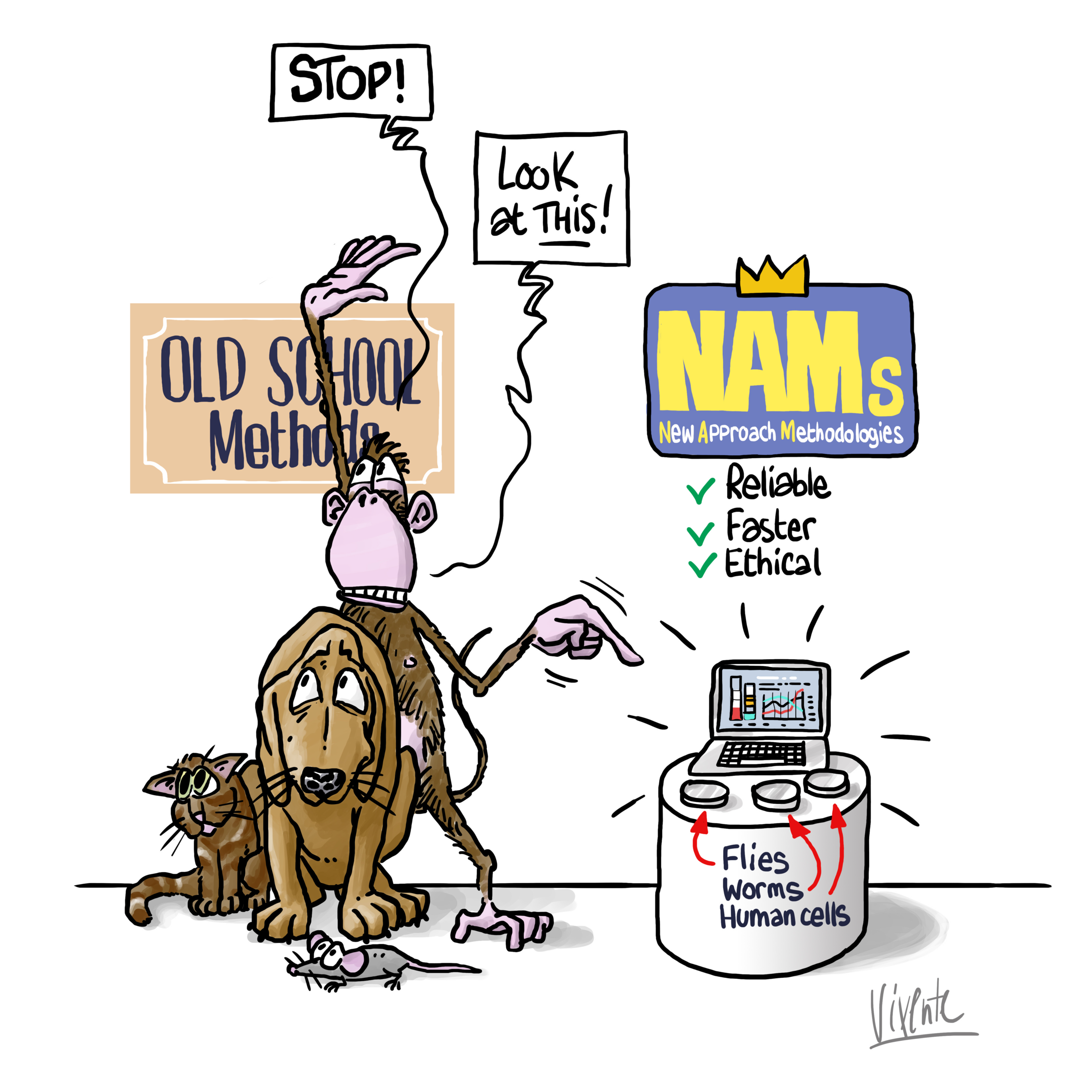
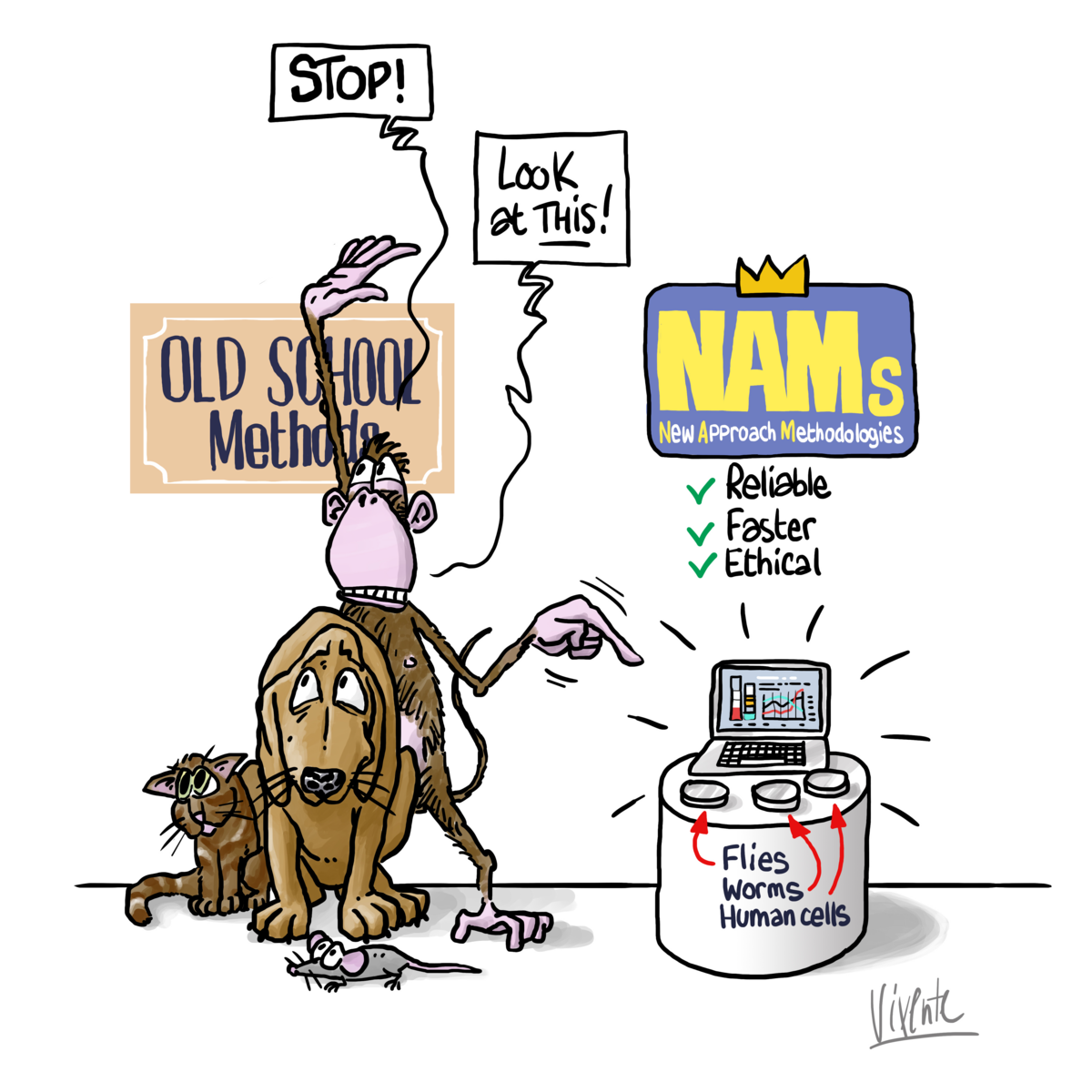
During the next Plenary session in Strasbourg in mid-September, Members of the European Parliament (MEPs) will vote on an important text for the 10 million laboratory animals used yearly in the European Union (EU). The motion for a resolution calls for an Action Plan to facilitate the transition to innovation without the use of animals in research, regulatory testing and education.
The EU is currently funding three research projects aiming to advance the safety assessment of chemicals without the use of animal testing: PrecisionTox, ONTOX and RISK-HUNT3R, led respectively by the University of Birmingham, Vrije Universiteit Brussels and the Leiden University, have formed the ASPIS cluster which gathers 70 research organisations and will receive 60 million € over the next 5 years to develop ethical solutions for the advancement of regulatory testing.
The Coordinators of the projects, Professor John Colbourne, Prof. Mathieu Vinken and Prof. Bob Van de Water, have joined their voices to support the EP initiative in a Statement so that this Motion for resolution is adopted and eventually translated into the EU legislative framework.
As a matter of fact, the European Commission announced a zero-pollution ambition in the Green Deal. The Chemical Strategy for Sustainability is expected to result in significant revisions of the EU legislative framework, such as the Cosmetics Regulation and the REACH Regulation, to deliver “a toxic-free environment “.
While toxicity testing on animals is costly and highly time-consuming (up to the animal’s entire life span), the number of chemicals whose toxicity is still unknown is always growing. Should the right support, concrete steps and achievable timeline be not taken in the near future to accelerate the transition towards non-animal models, not only the number of the animal experimentations will increase, but the promising ambition of the EU could turn into a backlog of new chemicals stuck in the review processes.
The coordinators state “The Motion for Resolution of the European Parliament is timely to accelerate this transition and meet the EU ambition to lead on the next generation for risk assessment in Europe and worldwide. “
Alternatives to animal testing, also known as New Approach Methods (NAMs), promise to fill the knowledge gap that challenges risk assessors and regulators with faster, more reliable and ethical solutions by enabling to better identify, classify and ultimately remove hazardous substances from the environment.



